The world of medicinal chemistry is experiencing a surge of excitement around peptides, peptidic, and semi-peptidic macrocycles as innovative beyond-rule-of-5 modalities. These structures are successfully unlocking previously "undruggable" targets.
However, linear and cyclic peptides face several challenges that limit their potential in optimisation campaigns, including:
- Rapid metabolism in blood, tissue, and the GI tract
- Fast excretion via liver and kidneys
- Low target specificity due to conformational flexibility
- Unwanted immunogenic responses
As the industry ventures deeper into Hit-to-Lead and Lead Optimization campaigns, these hurdles demand efficient medicinal chemistry strategies. Moreover, most DNA-encoded library (DEL) screening hits also require similar approaches.
The Role of Peptidomimetics in Drug Discovery
Peptidomimetic approaches bridge the gap between biologically active peptides and small-molecule drugs by enhancing key properties such as stability, selectivity, and pharmacokinetics.
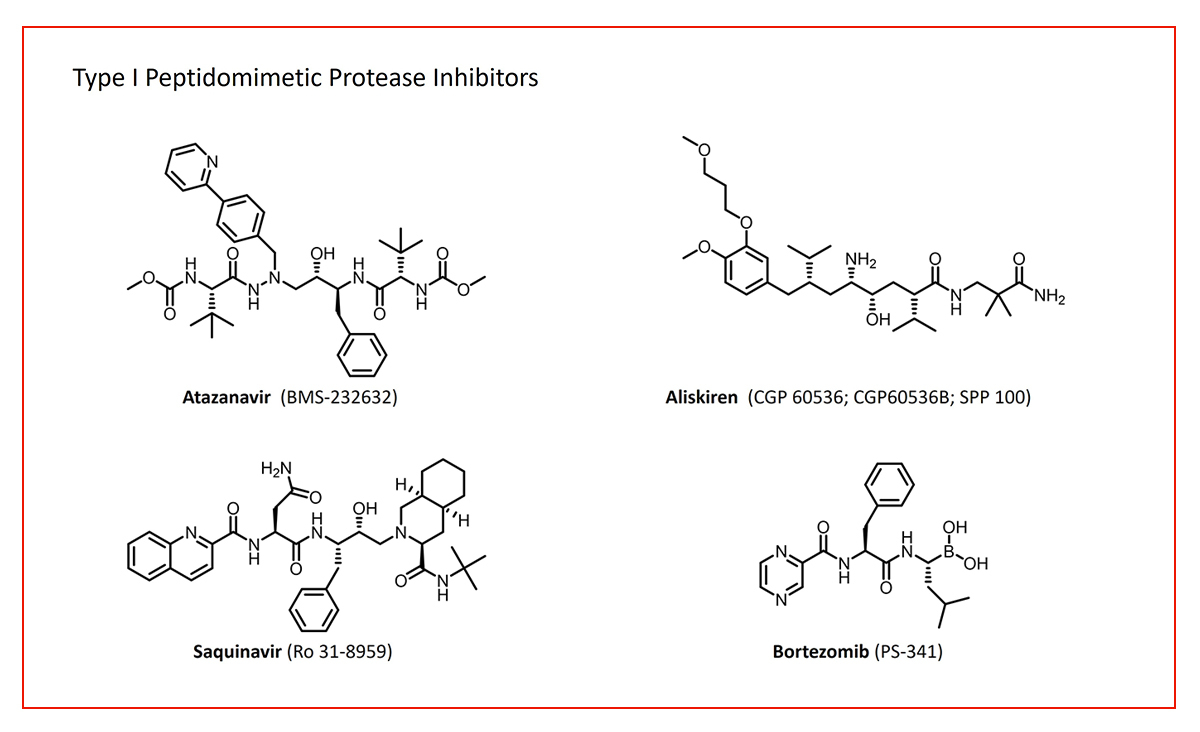
Type I peptidomimetics, such as inhibitors of various proteases, mimic the peptide backbone while incorporating sidechains derived from canonical amino acids (Fig. 1).
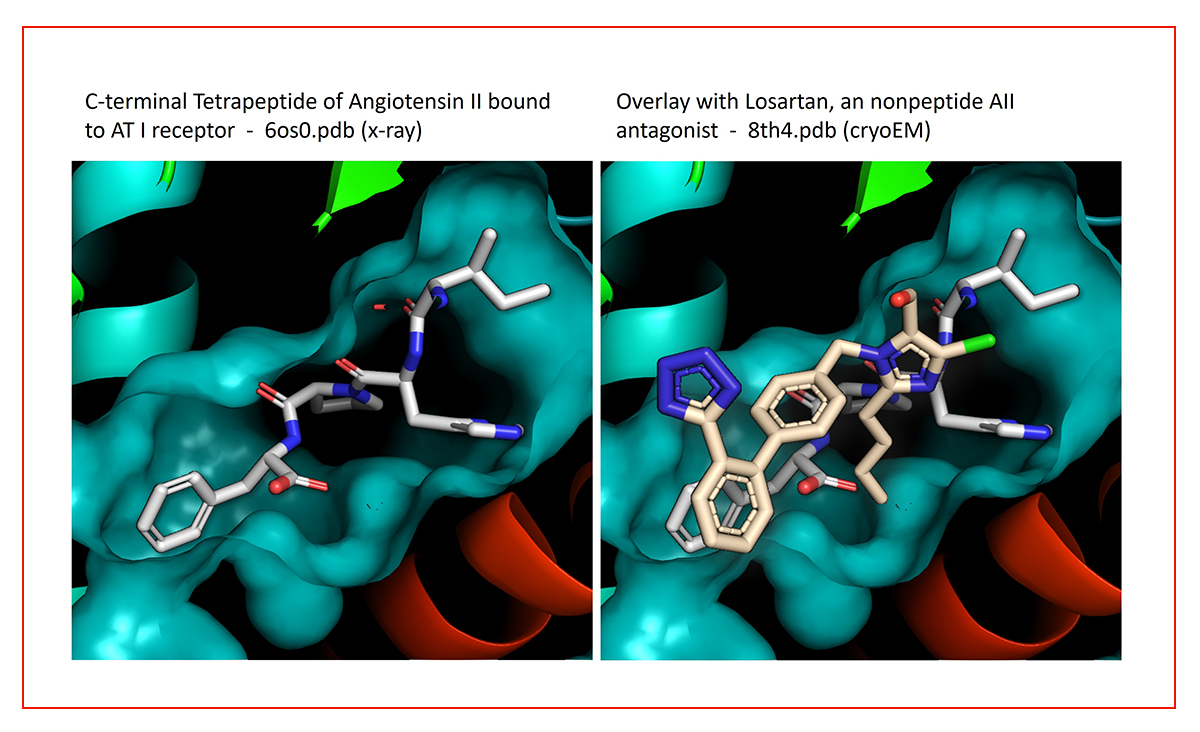
Type II peptidomimetics, like the Sartan class of non-peptidic angiotensin-II receptor antagonists, structurally mimic key interactions of the native peptide with little or no structural resemblance (Fig 2).
A Toolbox for Modern Medicinal Chemists
To guide the transition from peptides to drug-like molecules, medicinal chemists require an advanced toolbox for stepwise de-peptidization, ensuring:
- Stabilization of target-bound conformations
- Enhanced activity and selectivity
- Increased metabolic stability
- Optimized physicochemical & pharmacokinetic properties
At SpiroChem, we specialize in this intricate "molecular surgery." By seamlessly blending solid-phase synthesis with cutting-edge organic chemistry, we support hit-to-lead and lead optimization campaigns through a hierarchical toolbox approach, leveraging our expertise in:
- Amino acid scanning (retro-, inverse-, and retro-inverse analogues)
- Sidechain modifications for improved interactions
- Peptide bond replacements to enhance stability
- Secondary structure mimetics for optimal target engagement
A Toolbox for Modern Medicinal Chemists
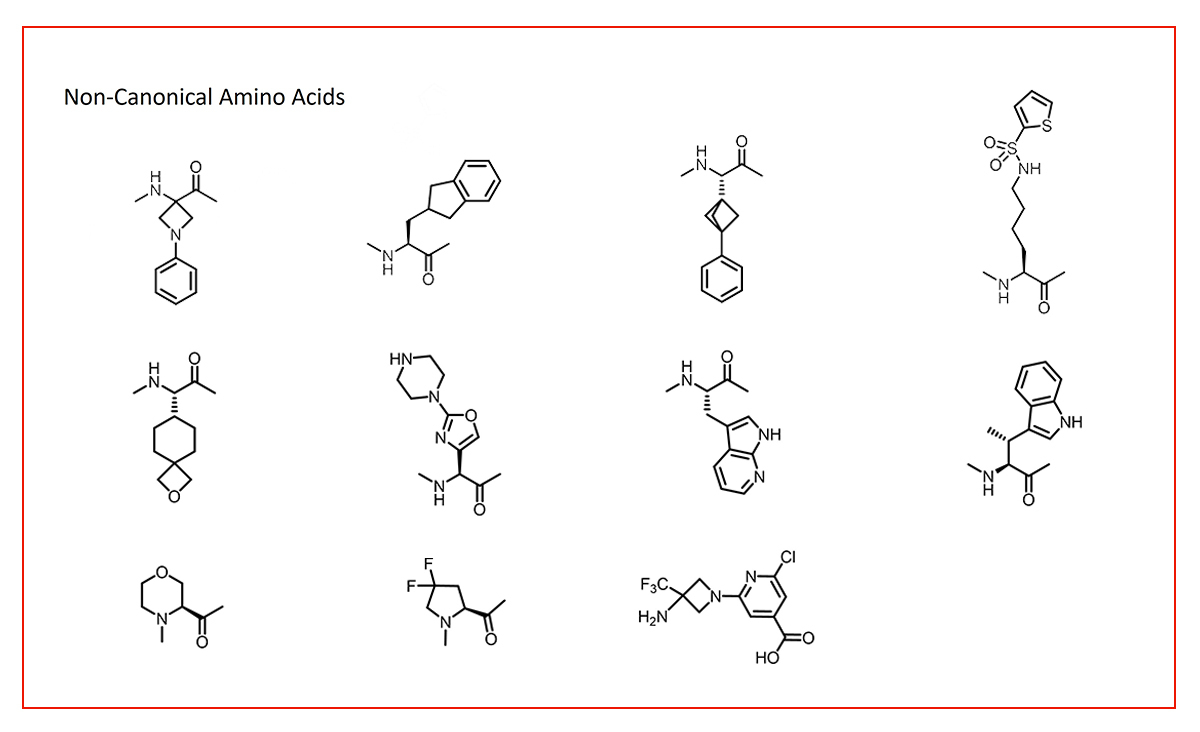
At SpiroChem, we have specialized in the design and synthesis of non-canonical amino acid analogues (Fig. 3) utilizing a broad spectrum of chemistries:
- Strecker and Bucherer-Bergs reactions
- Asymmetric alkylation
- Metal-catalyzed arylation
- Ionic and Radical additions
- Electrochemical modifications
Stay Tuned
Our next post will explore peptide bond and dipeptide isosteres, and secondary structure mimetics, further pushing the boundaries of medicinal chemistry.

























